
Nobuhiro Handa
Pharmaceuticals and Medical Devices Agency, Japan
Title: Nationwide registries associated with cardiovascular medical devices in Japan
Biography
Biography: Nobuhiro Handa
Abstract
The accumulation of real world data associated with medical devices, or a medical device registry plays an important role in promoting the development of medical devices. It is desirable to utilize registry data for pre-approval review as well as postmarketing
safety measures. However, data has often been collected without compliance with a good clinical practice. Furthermore, the registry may not include all data elements required for demonstrating the effectiveness and safety of medical devices. As such, it may not meet the regulatory requirements for pre-market review. Despite these limitations, there are many national registries, such as orthopedic total hip joint registries in multiple countries that have successfully detected the increased revision rate of metal on metal hip implant. In addition, there are several national registries in the European Union, US and Japan that include registries for artificial heart placement, trans-catheter aortic valve replacement and devices for peripheral arterial diseases. If registry is nationwide, it is an unbiased real world data for the country. The outputs of the analyses can be compared with registries in other countries if the findings are consistent between countries or not. The current issues of registry management include ensuring a wide patient coverage, involvement of regulatory authority, implementing rules of data utilization by the manufacturer, regulators and the academic sector, and obtaining sustainable funding for registry management. In the presentation, we focus on the eight existing nationwide registries associated with cardiovascular medical devices in Japan and discuss their characteristics including data quality, data utilization and financial sustainability. Some of them were utilized for regulatory purposes. Early participation of regulators for developing a national registry and a system that assures accuracy of data seems to be a key element for their regulatory applications based on Japanese PMD-ACT. The cooperation between the academic sector, industry and regulatory bodies is essential for utilizing registry data efficiently.
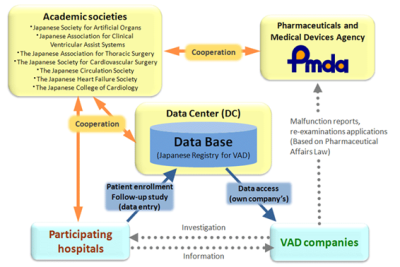
Figure 1: Registry framework of Japan mechanically assisted circulatory support

